In-vitro Microbiological Tests for the Characterization of Test Compounds:
- Minimum Inhibitory Concentration (MIC) assays determine the lowest concentration of an antimicrobial agent that prevents visible growth of a microorganism.
- Minimum Bactericidal Concentration (MBC) and Minimum Fungicidal Concentration (MFC) assays determine the lowest concentration of an antimicrobial agent required to achieve bactericidal and fungicidal killing, defined as a 99.9% reduction in the initial inoculum.
Emery Pharma routinely provides antibiotic susceptibility testing for the following methods according to Clinical and Laboratory Standards Institute (CLSI) guidelines. For more information, see our MIC guide in the resources section:
- broth micro- and macro-dilution (see illustration below)
- disk diffusion
- agar dilution
Test articles can be natural or synthetic, mixtures or purified. Hundreds of bacterial strains, including multi drug-resistant clinical isolates, ESKAPE pathogens, and multiple fungal strains, are available in our inventory for immediate testing.
ESKAPE Pathogens |
|
| Species | Phenotype |
| E. faecium | Gram +ve, VRE |
| S. aureus | Gram +ve, MRSA |
| K. pneumoniae | Gram -ve, MDR |
| A. baumannii | Gram -ve, MDR |
| P. aeruginosa | Gram -ve, MDR |
| E. cloacae | Gram -ve, ESBL |
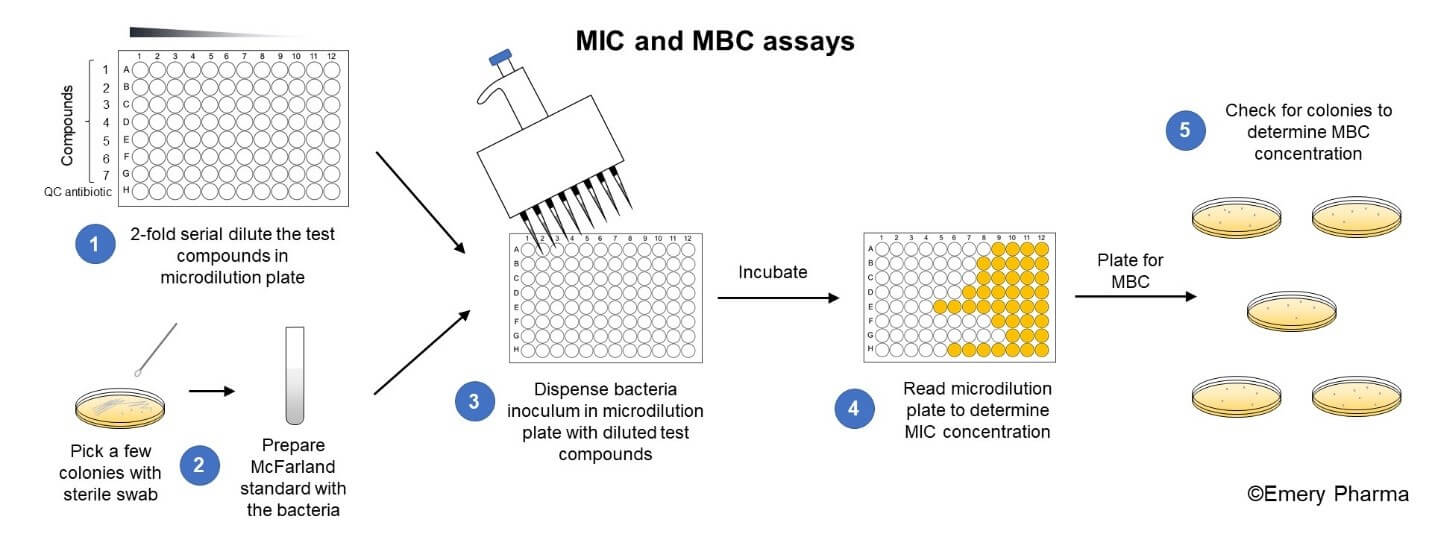
MIC and MBC Assays. To set up an MIC/MBC assay, (1) first prepare 2-fold serial dilutions of the test compounds (up to 7) and one quality control (QC) antibiotic in a microdilution plate. (2) Create the inoculum by taking a few colonies from an agar plate with a sterile swab, preparing a McFarland standard, and diluting the McFarland standard into media. (3) Dispense the inoculum into the microdilution plate with the serial diluted test compounds and incubate the microdilution plate. (4) Read the microdilution plate to determine the MIC value. (5) Plate a portion of each well on an appropriate agar media, incubate the agar, and check for colonies to determine the MBC.
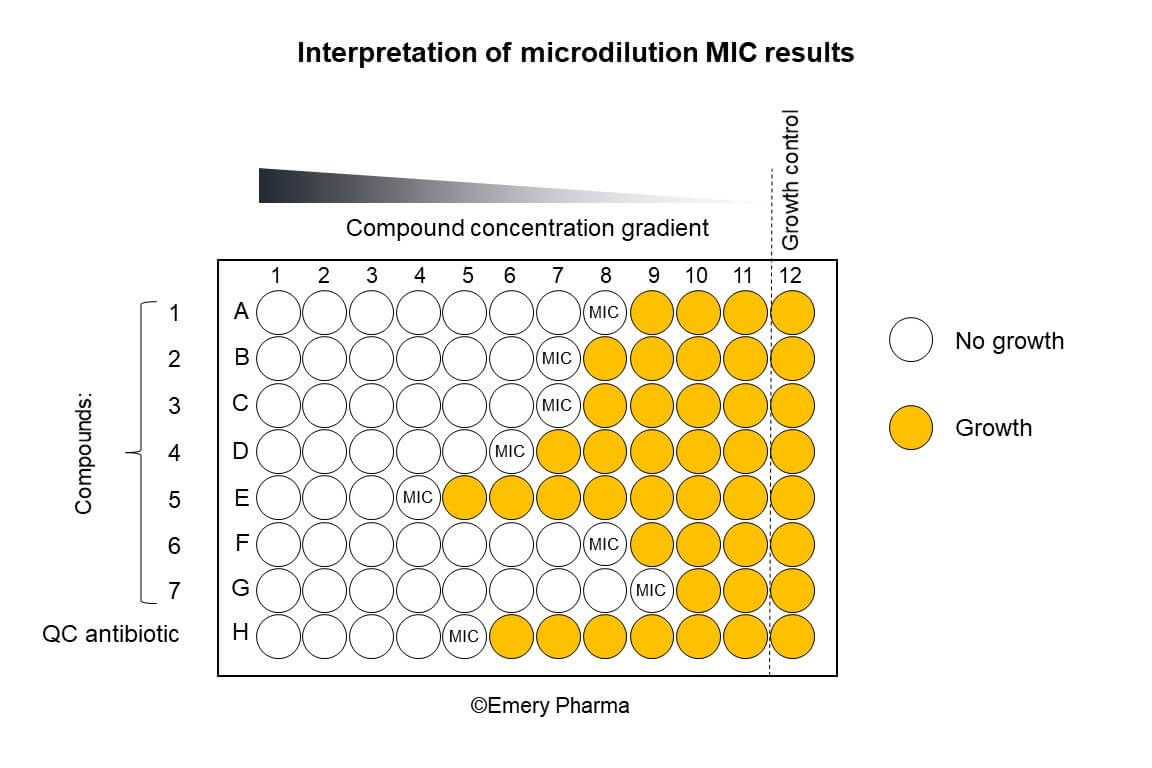
Interpretation of Microdilution Results. Depicted here is a typical MIC assay conducted according to CLSI microdilution guidelines. Up to 7 compounds and one quality control (QC) antibiotic are serially diluted from column 1 to column 11 of a 96-well microplate to form a concentration gradient. Column 12 serves as a positive growth control. In the illustration, “no growth” is represented by white circles and “growth” is represented by yellow circles. The MIC value is the lowest concentration of a compound/antibiotic at which no growth is observed.
References:
CLSI M100 Performance Standards for Antimicrobial Susceptibility Testing; Approved Standard-28th edition; January 2018
CLSI M07 Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically; Approved Standard-11th edition; January 2018.
CLSI M02 Performance Standards for Antimicrobial Disk Susceptibility Tests; Approved Standard-13th edition; January 2018.
CLSI M27 Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeasts; Approved Standard-4th edition; November 2017.
CLSI M38 Reference Method for Broth Dilution Antifungal Susceptibility Testing of Filamentous fungi; Approved Standard-3rd edition; November 2017.