Assignment of Complex NMR Spectra
Do you have a need for NMR services beyond basic 1-D proton spectra? Do you need expertise in analyzing and interpreting NMR data for discussions or presentations or publications? We can help by obtaining the specific 1-D and 2-D NMR experiments that will lead you to a high-level understanding of your compound. This will provide you with the information necessary to confidently make strategic decisions. We will be your partners in compound characterization and structural elucidation.
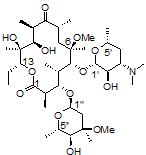
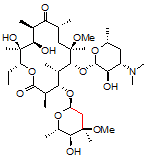
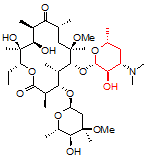
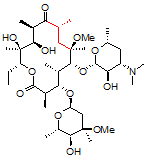
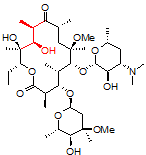
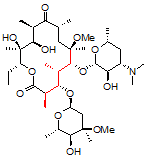
One challenging example of our services is shown below for the antibiotic clarithromycin. Clarithromycin is a semi-synthetic antibiotic which binds to the 50S ribosome, halting peptide synthesis in bacteria. A 14-membered macrolide, clarithromycin contains 38 carbons, 69 hydrogens, and 18 stereocenters, making NMR analysis a fun challenge for most organic chemists and a perfect molecule to showcase our capabilities in advanced NMR spectroscopy.
Obtaining ¹H NMR and ¹³C NMR spectra are straightforward on this compound, since the compound is very soluble, >10 mg/ml in CDCl₃. In this example, only 128 scans were required to obtain a ¹³C spectrum, which is a demonstration of how sensitive and quick these newer spectrometers have become; however, when characterizing compounds for peer-reviewed documents (e.g., ACS journals), we recommend many more scans to increase the signal-to-noise ratio. For a complex molecule such as clarithromycin, pro-R and pro-S hydrogens on methylene groups are often separated by a large chemical shift difference and cause problems in the assignment. As a demonstration of our capabilities, we will be using 2-D NMR spectroscopy as an easier way to assign the peaks.
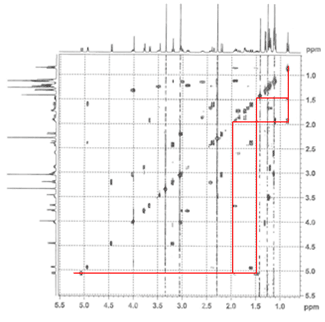
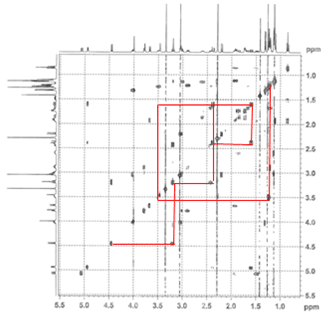
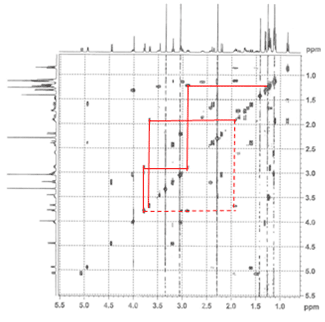
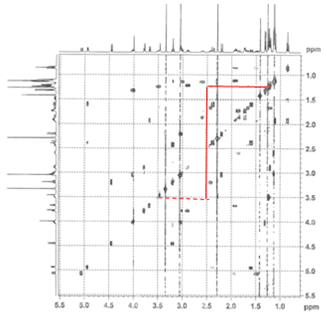
1. Correlation SpectroscopY (COSY) highlights 3-bond ¹H–¹H interactions and is useful for outlining different spin systems in a complex molecule. Clarithromycin has 7 different spin systems: C2–C5, C7–C8, C10–C11, C13, C1′–C6′, C1”–C2”, and C4”–C6”.
(a) The C13 spin system is easiest to identify due to the clear ethyl triplet at 0.85 ppm. Crosspeaks at (0.85, 1.49) and (0.85, 1.9) show the diastereotopic methylene hydrogens, and then cross-peaks at (1.49, 5.06) and (1.9, 5.06) indicate that the signal at 5.06 is the C13 hydrogen.
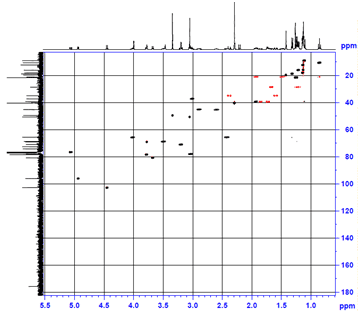
(b) The C1”–C2” spin system is second easiest to identify based on the assumption that the next two most downfield-shifted ¹H signals are the two acetal protons on C1′ and C1”. The C1”–C2” system is much simpler, involving only three protons, and as we can confirm on the HSQC spectrum (below), two of the protons are attached to the same carbon.
(c) As one of the larger spin systems, the C1′–C6′ spin system is more difficult to follow, but the COSY cross-peaks allow us to “walk” down the entire system, starting with the acetal proton at 4.45 ppm.
(d) The C7–C8 spin system is unique among the rest because there are no downfield-shifted protons.
The remaining three spin systems are similar in that all have the pattern CH₃–CH–CH(OH), with the C2–C5 spin system having two such units. As a result, several of the peaks are overlapping, and assignment is difficult but possible.
(e) The C4”–C6” spin system can be identified by the C5 proton, which is the most downshifted of all the remaining candidates. A multiplet at 4.02 ppm, it overlaps with an –OH signal, which we can determine from the HSQC (below).
(f–g) The C2–C5 spin system is differentiated from the C10–C11 spin system by the fact that it has an ester at one end (C1) rather than a ketone (C9). The two signals at 2.42 ppm and 2.89 ppm can be assigned to H10 and H2, respectively, and following the COSY cross-peaks helps us assign the final two spin systems.
The dashed lines show small cross-peaks which are only visible on a zoom-in.
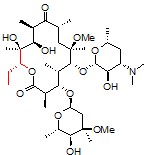
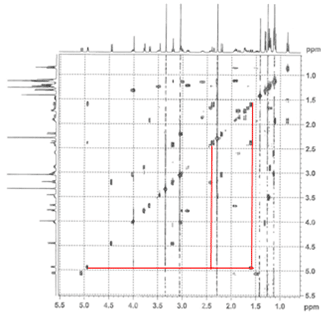
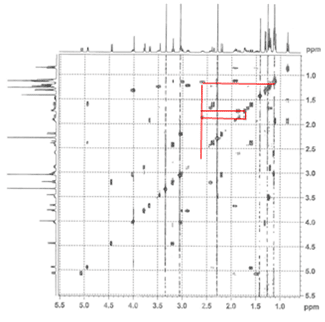
2. Heteronuclear Single Quantum Coherence (HSQC) spectroscopy highlights 1-bond ¹H–¹³C interactions (contrast to COSY, which highlights 3-bond ¹H–¹H interactions). Having assigned the ¹H NMR spectrum, we can now assign the ¹³C NMR spectrum with a single experiment.
This particular HSQC is multiplicity-edited, which means that CH and CH₃ groups are phased opposite of CH₂ groups (much like a DEPT-135 experiment). This gives us additional information about the diastereotopic methylene protons, and, as referenced above, can be used in differentiating geminal from vicinal couplings in the COSY spectrum.
Organic chemists can often benefit from the use of 2-D NMR techniques. In particular, 2-D spectra can often help resolve bunched peaks (e.g., the methyl region from 1.0–1.2 ppm), as well as unambiguously assign closely related peaks (e.g., acetylation of the 2′-OH vs. 4”-OH).
If you’re interested in 2-D NMR, complex NMR interpretation, and how it can help you with 1H NMR spectral assignment, contact Emery Pharma.
About the Author
Originally authored by Dr. Timothy Shiau. This article was reviewed and updated on May 28, 2025 by Dr. Ryan Cheu, current Director of Chemistry.