Calibration of the Variable Temperature NMR Thermocouple
In nuclear magnetic resonance (NMR) spectroscopy, especially when running variable temperature (VT) NMR experiments, getting the temperature right is important. Changes in temperature can affect chemical shift accuracy, reaction kinetics, or molecular motion. The challenge is that the temperature shown on most NMR instruments typically reflects the temperature of the airflow around the sample, not the actual internal temperature of the NMR tube. Calibrating the thermocouple helps ensure that your NMR data reflects the true experimental conditions that your sample is experiencing.
The variable temperature unit on Emery Pharma’s qualified 400 MHz NMR spectrometer uses a thermocouple wire to estimate the sample temperature. However, because the thermocouple does not directly contact the sample—it resides outside the NMR detection coils—there may be a discrepancy between the displayed (air) temperature and the real sample temperature.
To correct this, the thermocouple temperature can be calibrated using the temperature dependence of ¹H NMR chemical shifts in ethylene glycol (for high temperatures) or methanol (for low temperatures). In the example below, the chemical shift difference between the –OH and –CH₂– protons (denoted as Δ or D) in neat ethylene glycol was measured. The actual sample temperature (T in Kelvin) is then calculated using the following NMR calibration equation:
T = (4.637 – D) / 0.009967
In this sample spectrum, taken at room temperature, D = δ(OH) – δ(CH₂) = 1.665. This corresponds to a calculated sample temperature of 298.2 K. While the absolute chemical shift may drift slightly (due to the absence of deuterium in neat ethylene glycol, which prevents spectrometer locking), the difference in chemical shifts (Δ) remains stable and reliable for NMR temperature calibration.
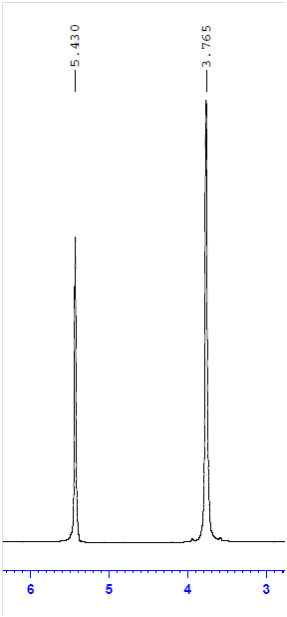
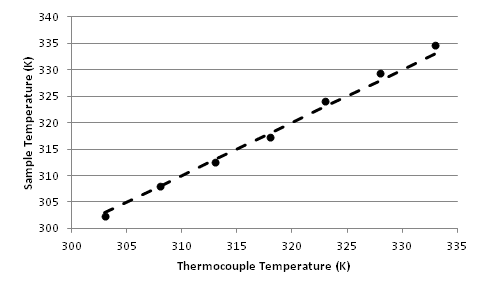
Plotted against the instrument's thermocouple readings, we can see that the thermocouple is usually within ±1 K of the actual sample temperature. However, for precision VT NMR experiments, we recommend calibrating the temperature using the measured internal temperature, not the thermocouple readout. It's also essential to ensure thermal equilibrium of all samples before initiating data acquisition.
For more routine VT NMR applications, such as 2D NMR spectroscopy, the built-in thermocouple temperature is generally accurate enough.
If you'd like to learn more about NMR calibration techniques, variable temperature NMR best practices, or advanced 2D NMR analysis, contact Emery Pharma today!
About the Author
Originally authored by Dr. Timothy Shiau. This article was reviewed and updated on June 2, 2025 by Dr. Ryan Cheu, current Director of Chemistry.
