Probiotic Dating Game

Probiotics, broadly defined as microorganisms that confer a health benefit when consumed or applied, have become a hot area of research and commercial activity in recent years. This is most evident by a quick stroll down the supplements aisle in any major supermarket or pharmacy, with dozens of products available for purchase. But while there is a great variety of probiotics, the composition of these probiotic products is equally as complex. A quick check of the labels show that most products are composed of multiple strains of probiotics, and often contain molecular ingredients (such as enzymes, botanical extracts, carbohydrates, etc.) as well. To ensure the consumer receives the full serving of probiotics and molecular ingredients indicated on the label, it is important to make sure that all of these ingredients—microbial and molecular—are biocompatible with one another.
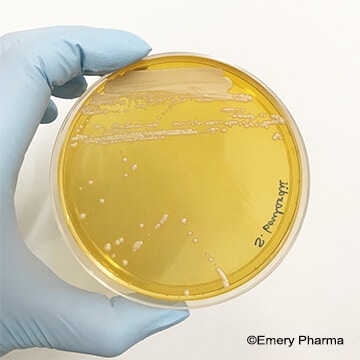
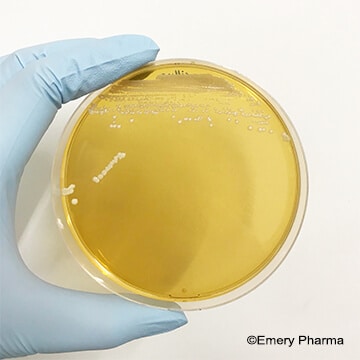
(Left: An image of Saccharomyces boulardii, a probiotic yeast, on yeast extract peptone dextrose (YEPD) agar. Right: An image of Lactobacillus plantarum, a probiotic bacteria, on De Man, Rogosa and Sharpe (MRS) agar.)
Many probiotics--such as Saccharomyces boulardii, Bacillus clausii, and some Lactobacilli spp.--naturally secrete antimicrobial molecules such as capric acid and bacteriocins [1-3], and these molecules may help protect the body from infection or modulate the immune system in humans. However, if the antimicrobial-producing probiotic is matched with a second probiotic, then care should be taken to ensure that antimicrobial molecules secreted by the first probiotic does not adversely affect the viability of the second probiotic. Additionally, although a probiotic may produce a natural antimicrobial molecule, higher than physiological concentrations of the same antimicrobial molecule may adversely affect the probiotic, which has evolved to tolerate the antimicrobial molecule within a limited range.
Similar considerations should be given when pairing probiotics with bioactive compounds. These compounds exhibiting inhibitory or microbicidal activity against a pathogenic species of microbe can just as likely do the same to a friendly probiotic microbe. In these cases, it is important to find the “center of balance” at a concentration at which a molecular ingredient can exert its activity against undesirable, pathogenic microbes while minimizing the impact on beneficial, probiotic microbes.
There are a number of assays for assessing the compatibility of probiotic + another probiotic or probiotic + other compound combinations, but one of the most common is the determination of probiotic density. Similar in principle to a time-kill assay, the probiotic is mixed with a secondary ingredient (either another probiotic or another compound) in suspension and the microbial density in colony forming units per milliliter (CFU/mL) is determined. This becomes the initial time point, or time zero. The probiotic mixture is then grown under the appropriate conditions and at a later time point, a portion of the probiotic mixture is sampled, and the CFU/mL density determined. If the CFU/mL in the probiotic mixture increased over time, then the secondary ingredient being paired with the probiotic is compatible with the probiotic under the conditions tested. Conversely, if the CFU/mL of the probiotic mixture decreased over time, then this is an indication that the secondary ingredient is likely incompatible with the probiotic under the conditions tested.
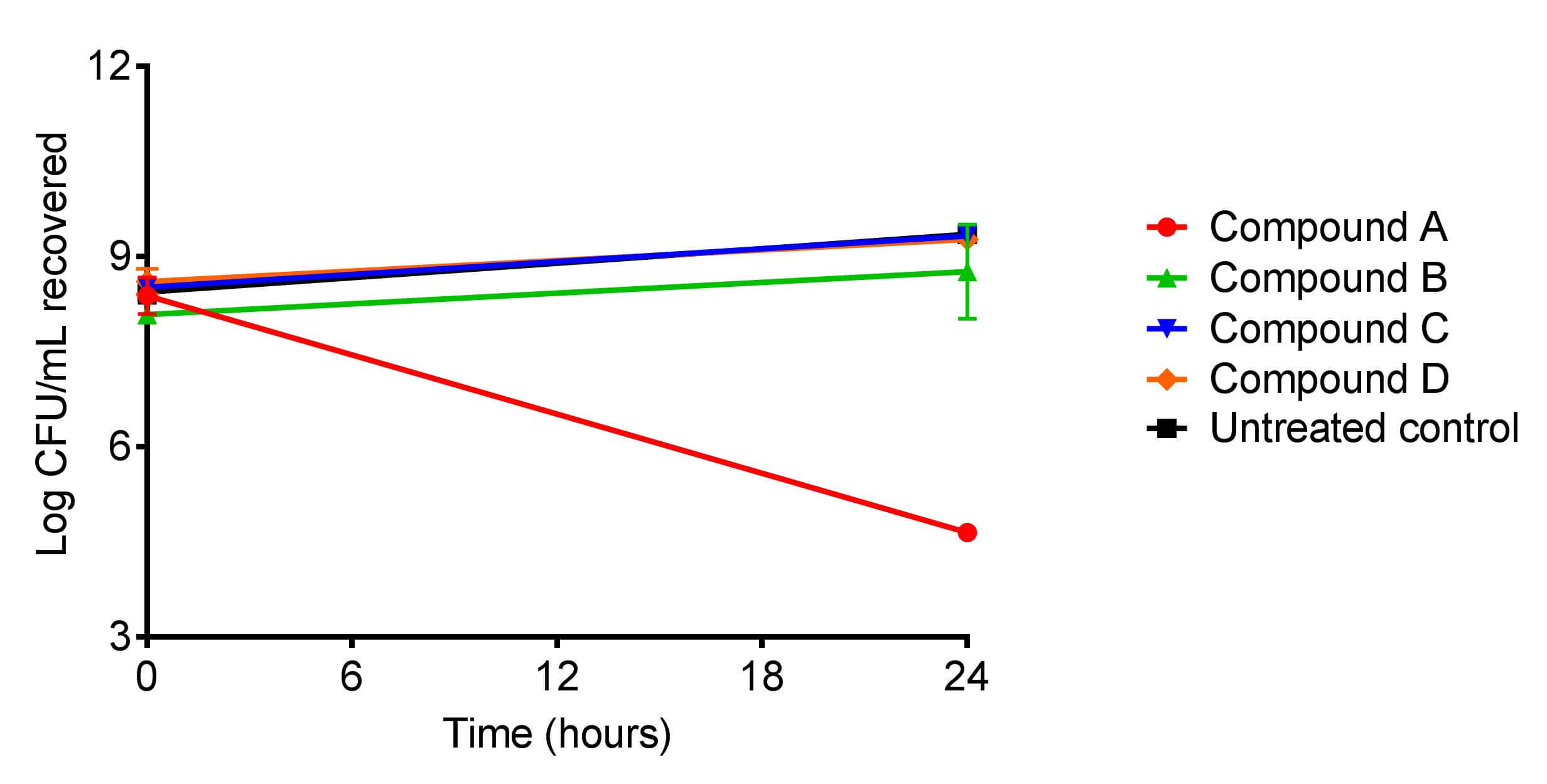
(A sample probiotic compatibility assay result. The effect of four different compounds on the growth of Lactobacillus acidophilus was assessed. Compound A has an inhibitory effect on the growth of L. acidophilus. In contrast, compounds B, C, and D have no adverse effect on the growth of the probiotic and L. acidophilus treated with these three compounds grew at a rate similar to the untreated control.)
In some ways, probiotic compatibility testing is a bit like a dating game show: the probiotics are seeking a compatible partner, and it is the scientist’s role as the host to devise the best questions in the form of assays to find the perfect match. At Emery Pharma our scientists have extensive experience working with probiotics and have successfully helped our clients bring their probiotic products to market. If you are interested in developing a new probiotic, contact us here and let us be your probiotic match-maker!
References:
- Capric Acid Secreted by boulardii Inhibits C. albicans Filamentous Growth, Adhesion and Biofilm Formation. Murzyn A, Krasowska A, Stefanowicz P, Dziadkowiec D, Łukaszewicz M. PLoS One. 2010 Aug 10;5(8).
- Bacillus clausii probiotic strains: antimicrobial and immunomodulatory activities. Urdaci MC, Bressollier P, Pinchuk I. Journal of Clinical Gastroenterology. 2004 Jul;38(6 Suppl):S86.
- Role of Lactobacillus reuteri in Human Health and Diseases. Qinghui Mu, Vincent J. Tavella, Xin M. Luo. Frontiers in Microbiology. 2018 Apr 19;9(757).
About the author:
Dr. Janet Liu is currently the Associate Director of Cell and Microbiology and is responsible for R&D and cGMP/GLP projects involving antimicrobial susceptibility testing, antimicrobial synergy, biofilm testing, time-kill assays, and cytotoxicity testing. Dr. Liu received her Ph.D. in Biomedical Sciences from the University of California, Irvine. She received her B.S. in Biological Sciences from the University of California, Davis.