Botanicals drugs, defined by the US FDA as “products from plant materials, algae, macroscopic fungi, and combinations thereof”, have been used in traditional medicines for thousands of years. Despite their long history, it was only in the last two decades that rigorous scientific research and development of botanical drugs have gathered pace. Drugs such as artemisinin, derived from the sweet wormwood plant, was recognized by the 2015 Nobel Prize in Physiology or Medicine, exemplifies the promise of botanical drugs.
Emery Pharma has extensive experience with all aspects of botanical drug discovery and development. We are happy to be the partnering lab for clients developing botanical drug products. Please contact us today to learn more about how we can collaborate.

Benefits of Botanical Drugs
Untapped Potential. Nature has created a combinatorial library of botanical ‘natural products’ for the benefit of humanity. While many botanical drug development groups focus on isolating and testing a single or a handful of compounds from botanical sources, whole botanical material consisting of a complex mixture of molecules is another important formulation that often go untapped for drug development. Recent FDA guidance on botanical drug development has jump-started interest in the field.
Multifactorial Activity Profile. Traditional mono-drug products generally have a single activity pathway and hence indication. Whereas a botanical specimen due to the presence of numerous biologically active molecules will entail multifactorial activity. As an example, the botanical drug substance, developed by "Company X" in collaboration with Emery Pharma, is a multifactorial botanical drug that has antimicrobial and wound healing activity, amongst other functions depicted below.
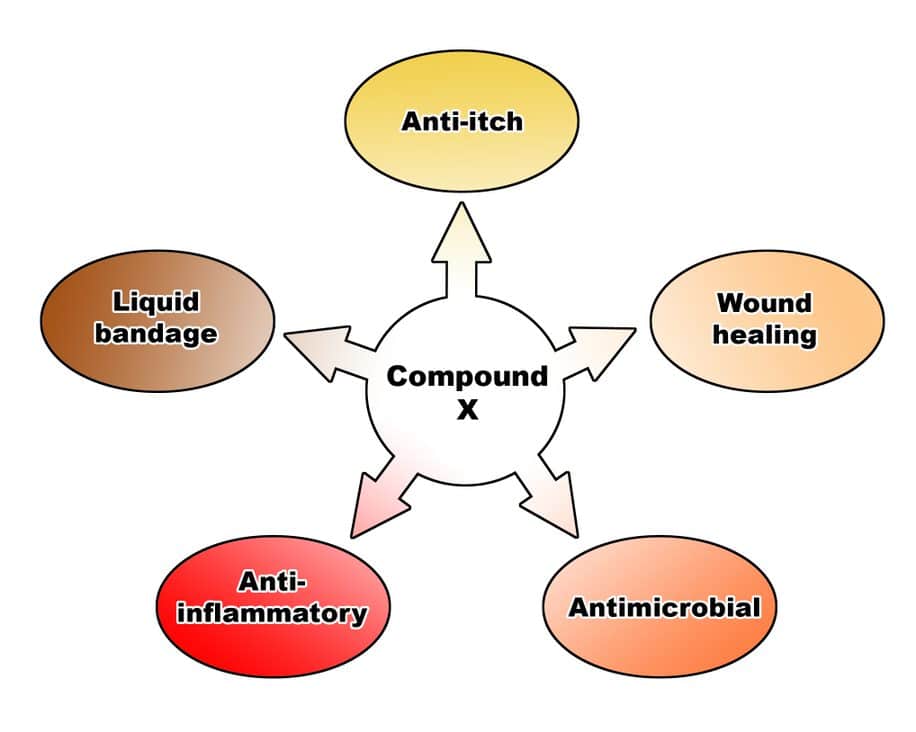
Exclusivity. Botanicals, by virtue of their complexity, have the added benefit of being difficult to genericize. Hence there are multiple opportunities for creating ‘trade-secrets’, as well as novel patents around the drug substance, its processing, formulation, etc.
Potential Expedited Regulatory Pathway. The US FDA has established a botanical drug guidance and recognizes the complexity of botanicals. Specifically, the fact that many of the typical requirements for mono-compound drug products cannot be readily performed (e.g., contribution of elements studies) mean faster regulatory approval.
Potential Accelerated Safety. Many botanicals have a long history of safe use in traditional medicines, which may be documented and reviewed in scientific literature. Existing scientific literature on safety may accelerate the safety review process, reducing the scope and financial burden for extensive safety studies.
Why Emery Pharma?
Track record. Emery Pharma has extensive experience in working with new botanical drugs and have been highly successful in spearheading multiple botanical drugs from conception, through discovery R&D, IND submission, and clinical trials.
The Ideal Team. To deal with unique challenges encountered in botanical drug discovery and development, we employ a unique team with multifaceted experience including traditional analytical chemistry, biochemistry, formulation science, natural products, and customized assay development.
The Complete Package. Scientists at Emery Pharma have engaged in multiple regulatory submissions for new botanical drugs and are prepared to assist Client partners across all stages of product development, including pre-clinical discovery, IND-enabling studies, pre-IND/IND meetings with the FDA, regulatory submissions, and commercialization. Along the way, we are happy to support our Clients with dissemination of scientific ideas for due-diligence purposes.
Listed below are a few key areas of consideration during a new botanical drug development program. We are well-versed with each of these challenges and can help you navigate this unique landscape!
Challenges with Botanical Drugs
Botanical Drug Substances Are Unique.
- Complex mixtures containing innumerable compounds in challenging biological matrices
- May lack a distinct active ingredient
- Mechanism-of-action may not be immediately obvious (likely synergistic interactions)
- History of substantial human use (in many instances, anecdotal)
- Batch-to-batch variability (biodiversity of the raw material)
Regulatory Challenges.
- Defining the “drug substance” and “drug product”
- Unique regulatory pathway for approval
- CMC considerations for biodiversity of raw (crude) material
- Challenges in establishing acceptance criteria for raw material, finished products, etc.
- Traditional ICH guidance does not apply for impurities/degradant testing
- Challenges in establishing stability-indicating criteria
- Difficulty formulating and testing formulations
- Walking a fine line between anecdotal safety data and expensive experimental toxicological assessment.
Requirements for Specialized Expertise.
- Personnel with diverse scientific backgrounds in natural products, customized assay development, bioanalysis, and formulations
- Specialized experience in regulatory submissions for botanical drugs, such as pre-IND briefing documents
- Navigating FDA pre-IND/IND meetings, responding to FDA requests and audits
- Education and communication with funders, and supporting due-diligence activities
We look forward to collaborating with you on your next botanical drug!