R&D and GLP/cGMP NMR Services
What is NMR?
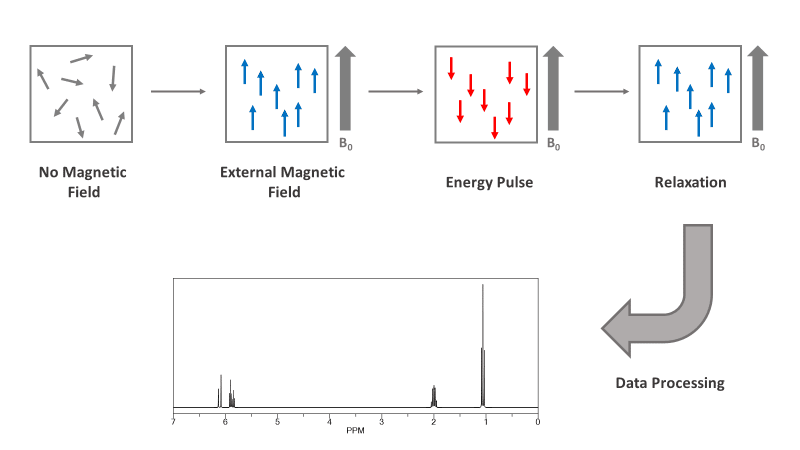
Nuclear Magnetic Resonance (NMR) spectroscopy is an analytical chemistry technique used to determine the molecular structure of a sample. This is done by analyzing the chemical environment of a selected nucleus. When nuclei are placed into a magnetic field, their magnetic moment (spin) becomes aligned with the magnetic field. NMR uses a pulse of RF (radio frequency) energy to deflect the nuclei. When the energy is removed, the nuclei relax back to their original state and emit an electromagnetic pulse. A coil inside the NMR receives this RF pulse, and a computer transforms the signal into a spectral graph, which can be read by a scientist. Each nucleus relaxation translates into a peak in the NMR spectrum. Its location on the x-axis (chemical shift) and the multiplicity of the peak tells the scientist about its environment. The area of the peak corresponds to the number of nuclei experiencing the same environment.
NMR is a powerful tool in structural characterization. It can be used to determine how all atoms of a particular molecule are interconnected, thereby determining a molecule’s content and purity.
Our NMR variable-temperature, tunable broadband probe can be set to virtually any nucleus that has a non-zero spin, including hydrogen, carbon, silicon, fluorine, chlorine, nitrogen, phosphorous, and more. One-dimensional and two-dimensional NMR experiments provide full characterization of known molecules and full structure elucidation of unknown compounds, such as impurities found in pharmaceutical products or metabolites from the biological breakdown of an active pharmaceutical ingredient (API).
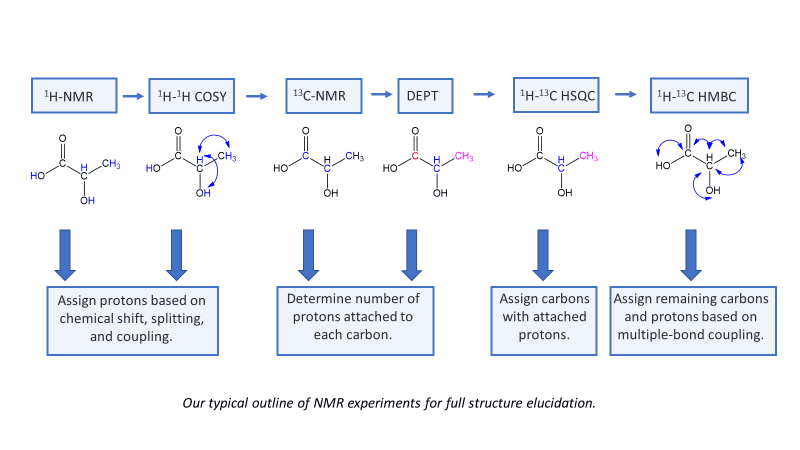
Thymidine is used in the following examples of common NMR experiments.
1-Dimensional NMR Experiments
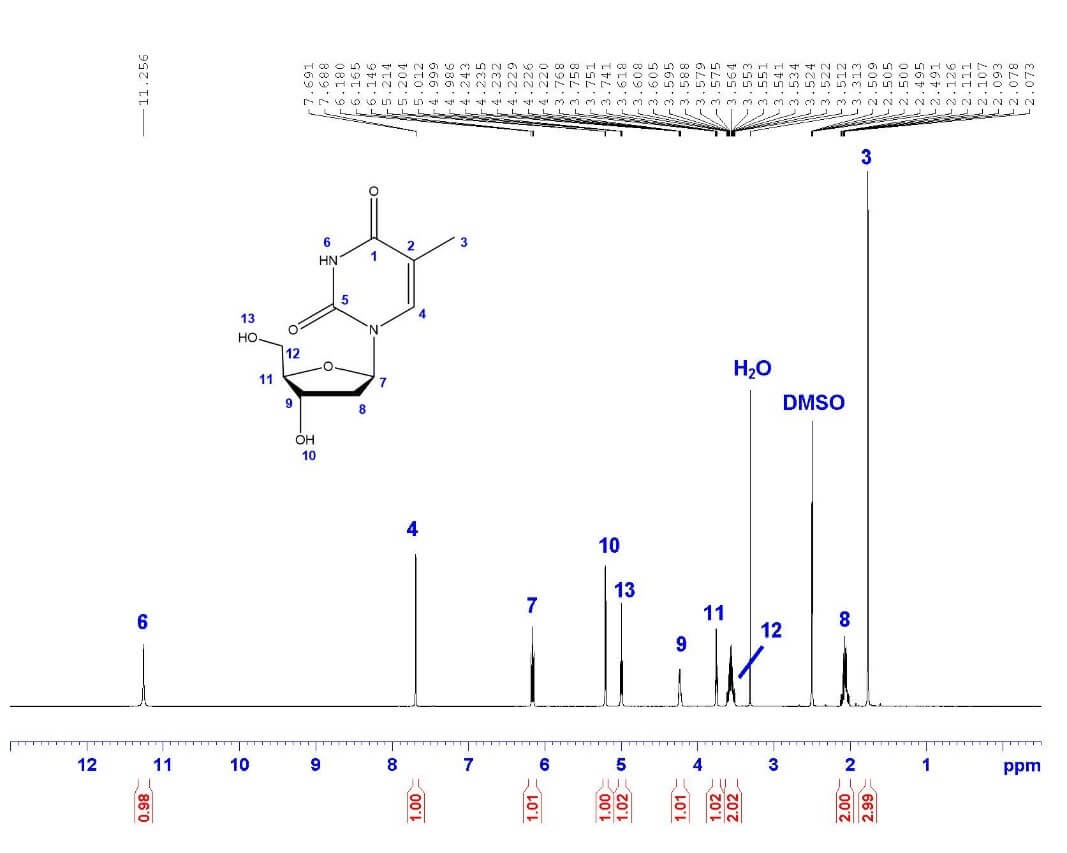
¹H NMR
The first step in structural characterization is a one-dimensional hydrogen NMR. We almost always start by acquiring a 1H-NMR to get a rough idea of the concentration and purity of the sample. It is also the fastest NMR experiment to run.
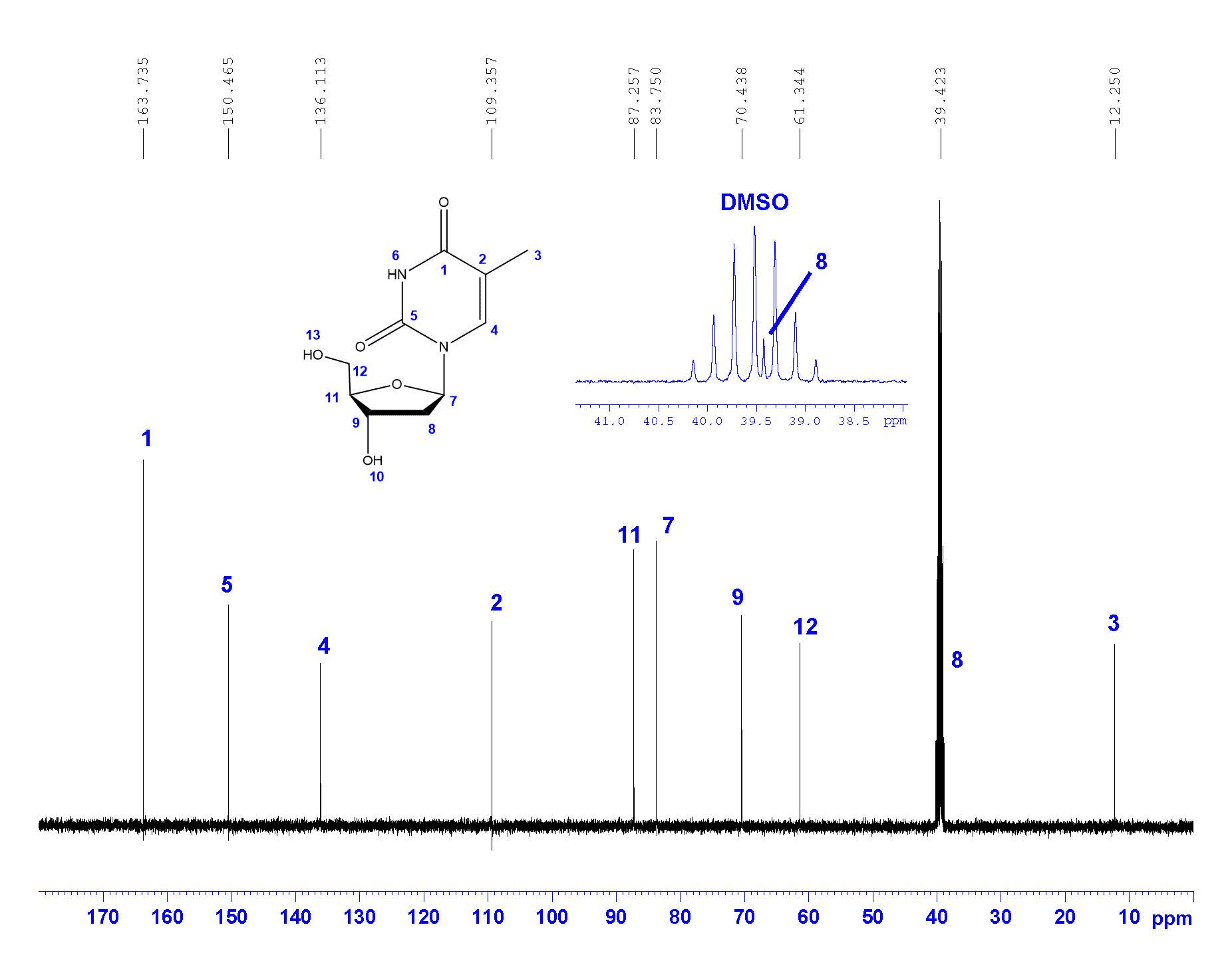
¹³C-NMR
Carbon NMR is a necessary step in full structural characterization. Usually, 13C-NMR alone does not provide enough information to assign the carbons in the molecule. Two-dimensional techniques are often necessary to assign all carbons with confidence.
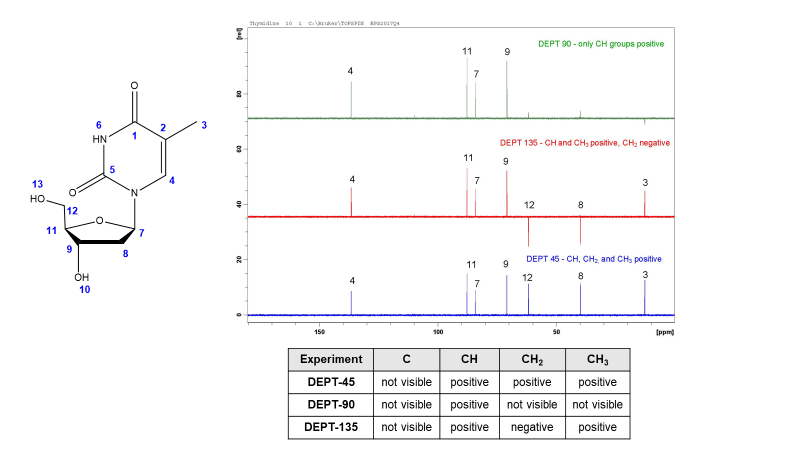
DEPT-45, 90, and 135
Distortionless Enhancement of Polarization Transfer (DEPT) experiments help assign carbon peaks by determining the number of hydrogens attached to each carbon. In complex molecules, DEPT and HSQC together are useful for confirming both carbon and proton assignments. There are 3 main DEPT experiments. Acquiring a carbon spectrum and several DEPT experiments will provide the number of hydrogens attached to each carbon atom.
2-Dimensional NMR Experiments
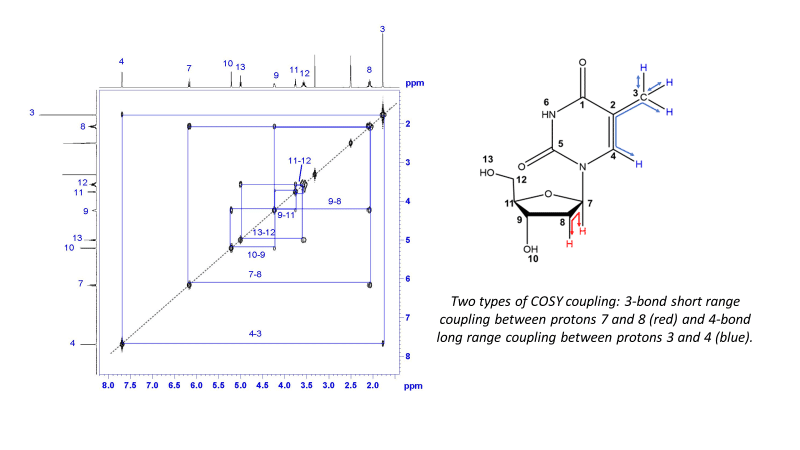
¹H-¹H COSY
1H–1H Correlation Spectroscopy (COSY) shows the correlation between hydrogens which are coupled to each other in the 1H NMR spectrum. In general, this indicates which hydrogen atoms are adjacent to another group of hydrogens. The 1H spectrum is plotted on both axes (2D). While 2-bond and 3-bond 1H-1H coupling is easily visible by COSY, long range coupling can also be observed with long acquisition times. The cross-peaks that are symmetric to the diagonal show the COSY correlations.
¹H-¹³C HSQC
1H–13C Heteronuclear Single Quantum Coherence (HSQC) Spectroscopy shows which hydrogens are directly attached to which carbon atoms. The 1H spectrum is shown on the top axis and the 13C spectrum is shown on the left axis. Only 1-bond correlations are observed by HSQC.
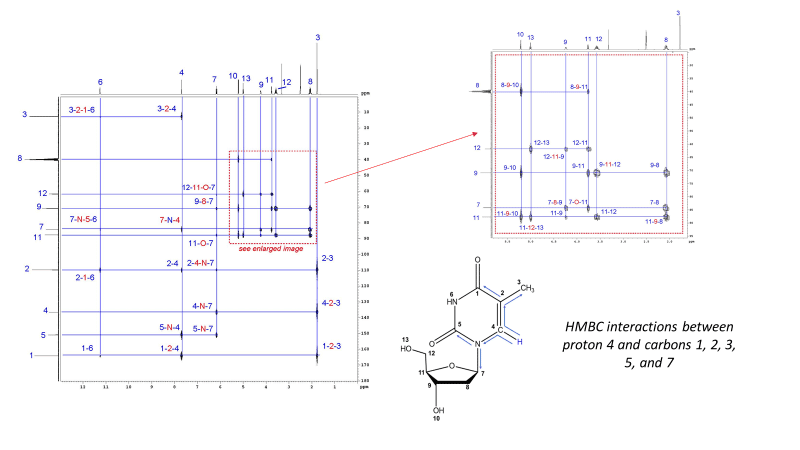
The experiments explained above are the most popular. For a detailed lesson on assigning protons and carbons in Thymidine by NMR, check out our blog. We also perform many more 1-dimensional and 2-dimensional experiments not discussed above, including Nuclear Overhauser Effect (1D NOE and 2D NOESY through-space correlations), multi-nuclear HSQC and HMBC, and more.