Antibody Drug Conjugates Q&A: A Novel Approach to Drug Delivery in Oncology
What is an antibody drug conjugate?
An antibody drug conjugate (ADC) is an innovative drug modality that consists of three main components: an antibody, a linker, and a drug, often referred to as a “payload” due to its cytotoxic nature. The antibody is a protein that binds to a specific antigen, or protein, which is expressed on the surface of tumor cells. The linker is a chemical bond that connects the antibody to the payload. The payload is typically a small molecule cytotoxic agent that can kill cancer cells once released inside the tumor cell.
What are the advantages of ADCs over conventional drug delivery methods?
Antibody drug conjugates offer several advantages over conventional oncology drug delivery approaches. First, ADCs can deliver a higher dose of a cytotoxic agent directly to tumor cells while reducing exposure and toxicity to normal cells. This is because the antibody acts as a homing device that guides the drug to tumor cells, while the linker acts as a gatekeeper that releases the drug only inside those targeted cells. This targeted delivery allows an ADC to reach a higher therapeutic index, which is the ratio of the effective dose to the toxic dose.
Second, ADCs can overcome some resistance mechanisms that tumor cells develop to evade conventional chemotherapeutics. An ADC can bypass barriers that often prevent drugs from reaching tumor cells, such as the blood-brain barrier, the tumor microenvironment, and cellular efflux pumps. Moreover, an ADC can target multiple biological pathways involved in tumor growth and survival, including apoptosis, angiogenesis, and cell cycle regulation.
What are the main challenges and limitations of ADCs in terms of finding the optimal combination of antibody, linker, and drug?
The development of effective antibody drug conjugates requires careful optimization of each component:
- The antibody should demonstrate high affinity, specificity, and internalization rate for the target antigen.
- The linker must balance stability in the bloodstream with the ability to release the payload inside tumor cells.
- The drug payload should exhibit high potency, solubility, and stability.
The overall formulation must also achieve a suitable drug-to-antibody ratio (DAR), which is the average number of drug molecules bound to each antibody molecule. DAR directly impacts both efficacy and safety. If the DAR is too low, the ADC may lose potency; if it is too high, the ADC may cause greater toxicity, raising safety concerns.
What are some stability and safety considerations?
An ADC must remain stable enough to maintain its integrity and biological activity during storage, transportation, and administration. However, ADCs are susceptible to degradation, aggregation, or deconjugation due to factors such as temperature, pH, light exposure, and enzymatic activity. These changes may compromise the quality, potency, and safety of the ADC. Therefore, rigorous monitoring and control of ADC stability and safety are critical throughout development, manufacturing, and clinical use.
Can you elaborate on the critical role of analytical services in the quality assurance of antibody drug conjugates (ADCs)?
Analytical services for ADCs are the cornerstone of quality assurance in oncology drug development. These services examine how ADCs behave under physiological conditions and provide key metrics such as serum stability and DAR measurements. These data points are essential for evaluating an ADC’s fitness for clinical applications. By offering insights into pharmacokinetics, stability, and structural integrity, analytical testing ensures that only ADC candidates with the most promising therapeutic profiles advance to clinical and therapeutic use.
In what way do serum stability studies enhance the quality evaluation of ADCs?
Serum stability studies provide critical insights into the long-term performance of antibody drug conjugates in the human body. By simulating physiological conditions, these studies assess how ADCs maintain structural integrity, potency, and therapeutic activity over time. This evaluation is vital for confirming an ADC’s safety, stability, and effectiveness throughout its shelf life and dosing schedule.
What are the current strategies for analyzing ADCs in biological matrices for pharmacokinetics (PK) and toxicokinetics (TK)?
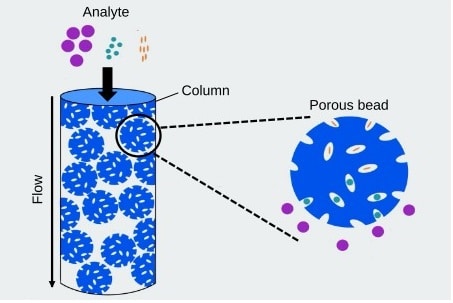
Analyzing ADCs in biological matrices for pharmacokinetics (PK) and toxicokinetics (TK) requires a multifaceted approach due to the hybrid nature of ADCs, which combine an antibody with a cytotoxic small molecule. Current strategies include advanced separation and detection techniques such as Liquid Chromatography Tandem Mass Spectrometry (LC-MS/MS), which is widely used for quantifying small-molecule payloads conjugated to antibodies with high sensitivity and specificity for both free and conjugated forms in biomatrices. Immunoassays, including enzyme-linked immunosorbent assays (ELISA) and electrochemiluminescence assays, are also commonly employed for detecting and quantifying the antibody component of ADCs. In addition, hybrid LC-MS/MS and immunoassay methods combine the strengths of both approaches, capturing an ADC or its components for subsequent LC-MS/MS analysis, thereby enhancing sensitivity and specificity in ADC pharmacokinetics and toxicokinetics studies.
At Emery Pharma, we have the expertise and advanced instrumentation, including LC-MS/MS and ELISA platforms, to perform the full range of studies essential for antibody drug conjugate development. Contact us online or call us at +1 (510) 899-8814 to learn how our team can support your ADC analytical and bioanalytical testing needs.
About the Author
Authored by Dr. Prajita Pandey, Associate Director of Chemistry.