Nitrosamine Impurity Found in Januvia®, a Popular Diabetes Medication
A Nitrosamine impurity known as Nitroso-STG-19 (NTTP), has been identified in Januvia®, a popular diabetes drug developed by Merck & Co. Januvia® (sitagliptin), is an oral medication used to treat Type II Diabetes. According to Bloomberg News, Januvia is Merck’s third best-selling drug with $3.3 billion in sales last year.
Emery Pharma is developing sensitive methods for detecting NTTP in Januvia® products and has launched an investigation into the root cause of NTTP formation utilizing a newly acquired Thermo Scientific High-resolution mass spectrometer (Exploris 240). Emery Pharma has extensive experience with nitrosamine impurities in other pharmaceuticals, such as Zantac (ranitidine), valsartan, and metformin.

NTTP belongs to the Nitrosamine class of compounds, which have been classified as probable human carcinogens. There is no available data to directly calculate the carcinogenic risk of NTTP, however, the FDA has used data from closely related nitrosamine compounds to calculate lifetime exposure limits for NTTP.
The FDA announced that in order to avoid a drug shortage and decreased access to the medication, the agency will allow distribution of sitagliptin containing NTTP up to 246.7 ng per day (which is above the acceptable intake limit of 37 ng per day). Merck has confirmed that NTTP has also been detected in batches of Janumet® and Steglujan®, other sitagliptin combination drugs.
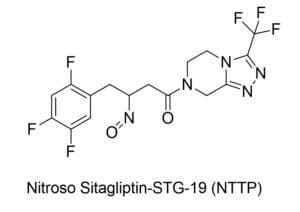
The structure of NTTP along with its prevalence in Januvia® and combination drugs, suggest that the impurity may be linked to the Januvia® molecule itself, and requires further investigation.
To learn more about our in-house methods or for inquiries regarding testing Januvia® products, please contact us.
For all inquiries, please contact:
info@emerypharma.com
Tel: (510) 899-8814
