A Fascination for the Human Microbiome for a Healthier Life
Trillions of microorganisms live in and on our body. It is estimated that there are 10-100 trillion microbes in a healthy human microbiome, outnumbering human cells by about 10 to 1. But they are not just mere passengers; in fact, they are a coalition of bacteria, fungi, and viruses playing important roles in our metabolism, immunity, and overall good health. In this blog, we will talk about the gut and skin microbiome, and how the human system depends on their presence.

Dysbiosis and the Gut Microbiome
95 percent of human microbes are found in the gut, so it makes sense that it is the most widely researched microbiome in the body. At the beginning of development and at birth, a newborn’s digestive tract is populated by microbes from the mother’s skin and vaginal canal. Interestingly, vaginally-born babies have a markedly different microbiome from babies delivered by a C-section. The differences in the microbiotas may be the reason why babies born vaginally generally have lower rates of infections and asthma [1]. As the child develops, the nature of gut microbiome is affected by a combination of environment, diet, and age. Foods with a high content of probiotics (for example, yogurt, apple cider vinegar, and miso soup) can give one’s body beneficial bacteria. On the other hand, processed foods such as sugar, gluten, etc. can negatively affect intestinal health; limited consumption of these foods is extremely beneficial [1].A healthy internal microbial ecosystem is diverse but stable. Any alteration of our microbial ecosystem causes us to enter a state of dysbiosis, or microbial imbalance. Dysbiosis generally occurs when there is an excess number of harmful bacteria or when there is a depleted number of beneficial bacteria caused by excess stress, unhealthy diets, parasite infections, or a variety of other factors. Numerous pieces of evidence have also shown the connection between antibiotics and dysbiosis. One study found that broad-spectrum antibiotics can cause a 30% reduction in intestinal bacteria, in addition to decreasing bacterial diversity. When an antibiotic regimen is concluded, the microbiota will attempt to return to its initial state, but it will never be exactly at the state it was before [2].

There is evidence that shows that dysbiosis has a role in other chronic conditions, including inflammatory bowel diseases such as Crohn’s disease and ulcerative colitis, coeliac disease, obesity, asthma, and HIV disease progression [3]. Evidence even suggests that dysbiosis has a more significant role than host genetics in causing diseases. On top of that, opportunistic pathogens can foster in a depleted gut microbiome and leave a tremendous impact. Two common pathogens are Helicobacter pylori and Clostridium Difficile [4 – 5]. The natural microbiome in the gut not only fight pathogenic bacteria but also, have health benefits such as improved metabolism. Lactobacillus, for instance, is a beneficial bacterium that helps digestion by turning food into usable nutrients by its secretion of lactase and vitamin K. Not only do these microorganisms impact our physical health, but also impacts our mental health. In fact, some neurochemicals are produced by bacteria; for example, serotonin, a chemical that makes an individual feel happy, is mainly produced in the gut by bacteria [6].
Dysbiosis and the Skin microbiome
There is a strong connection between dermatological disorders and an imbalance of the skin’s microbiome. Dysbiosis of the skin microbiome is linked to several skin conditions. The most common is the Atopic Dermatitis (AD), also known as Atopic Eczema, which is an inflammatory skin condition characterized by itchy, swollen, and cracked skin. The onset of AD occurs when there is a depletion of commensal microorganisms, such as S. epidermidis and S. hominis, due to overuse and misuse of antibiotics and antiseptic soaps. The role of S. epidermidis and S. hominis on the skin is to produce bacteriocins that the inhibits the growth of pathogenic bacteria, such as Staphylococcus aureus [7 - 8]. To counter this skin condition, some researchers are utilizing probiotic bacteria such as Lactobacillus brevis to enrich and promote the growth of S. epidermidis to improve the skin health [9].
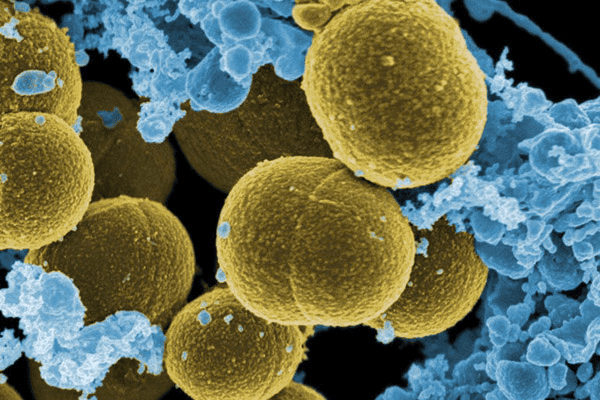
Electron microscopic image of pathogenic S. aureus bacteria found on skin (Photo courtesy - NIAID)
The skin microbiome also plays a key role in acne, a skin condition characterized by severe pimples, blackheads, papules, cysts, and nodules, that is caused by Propionibacterium acnes. Research has shown that certain strains of P. acnes support metabolism and are considered important for healthy skin. However, other research suggests that there are also other less beneficial strains of P. acnes which are more likely to trigger skin damage and the conditions associated with acne. [10].
It has also been suggested that the microbiome on the scalp is being linked to severity of dandruff. The scalp microbiome is important in preventing skin barrier disruption, controlling epidermal cellular proliferation and differentiation, and balancing shifts in sebum composition. Imbalance in scalp microbiome results in the increase of Staphylococcus and Malassezia fungi, and thus depletion of beneficial Propionibacterium, leading to increases in dandruff severity [11].
Microbiome Innovations
The market for microbiome-related research and development has skyrocketed, with several companies and individuals standing out with innovative ideas. To name a few of the remarkable innovations by emerging companies in the field of microbiomes:
Eligo Bioscience is developing a programmable biotherapeutics platform to treat bacteria related diseases and improve overall health. The new generation of drugs from Eligo Bioscience will essentially be a biological nanobot made up of DNA and protein capable of delivering a customized therapeutic payload to targeted pathogenic bacteria. Upon finding their target bacteria, these nanobots either kill or transiently transform into drug-producing agents to ameliorate the disease [12].
Kallyope Inc. is utilizing gut-brain axis functions to improve human health. Kallyope Inc. technology integrates cutting-edge technologies in microbiome research to provide an understanding of gut-brain biology. This will advance transformational therapeutics and consumer products to improve human health and nutrition [13].
Xycrobe Therapeutics Inc. is developing cutting-edge skin care products to treat inflammatory skin diseases such as acne, psoriasis, and dermatitis. Xycrobe’s microbial-delivery-system platform technology will be capable to distribute an array of biotherapeutics directly to target tissues, thereby substantially improving health and quality of life [14].
Evolve BioSystems is solving newborn gut dysbiosis. Evolve’s product, Evivo™ (activated B. infantis EVC001, ActiBif®) with MCT Oil, is the first and the only clinically proven probiotic to work with breast milk to restore a baby's gut microbiome to its natural state. It is available as a single-use administration for infants in a hospital setting. Administering Evivo with breast milk is clinically proven to allow B. infantis to thrive and dominate the infant gut microbiome, while also reducing relative populations of potentially harmful bacteria, such as E. coli, Clostridia, Staphylococcus (Staph), and Streptococcus (Strep) [15].
Enterome Bioscience is using gut microbiome research to treat inflammatory bowel diseases and immuno-oncology diseases. Enterome Bioscience has developed a first-in-class FimH blocker for the treatment of Crohn’s disease and EO2315, a novel immuno-oncology candidate in patients with aggressive brain cancer [16].
Rebiotix Inc.’s Microbiota Restoration Therapy™ (MRT) platform delivers healthy, live, human-derived microbes into a sick patient’s intestinal tract to treat diseases. Their MRT product, RBX2660 is currently in Phase 2 randomized, placebo-controlled, double-blind study for treating recurrent Clostridium difficile infection [17].
At Emery Pharma, we have extensive experience in testing bioactive ingredients that promote the growth of beneficial bacteria and inhibit the growth of pathogenic bacteria, such as the MIC and time-kill assays. Additionally, we the capability of testing the safety of novel adjuvants often used in microbiome research. Let us know how we can collaborate. Everything we do at Emery Pharma focuses on our mission of “helping to save lives and save the environment”.
About the authors
Leila Bonakdar was a Research Associate at Emery Pharma and she has a bachelor’s degree in Biology from the University of California, Berkeley, CA. Leila is currently a Research Intern at Alcon, a Novartis Division, Dallas, TX, USA.
Aava Farhadi was a Research Associate and an Intern at Emery Pharma and student at Northgate High School, Walnut Creek, CA, USA.
Neeku Mahdavian is the Business Development Manager at Emery Pharma and Neeku has a bachelor’s degree in Biology from San Francisco State University, San Francisco, CA, USA.
References:
- Carding, Simon, et al. "Dysbiosis of the gut microbiota in disease." Microbial ecology in health and disease 26.1 (2015): 26191.
- Francino, M. P. "Antibiotics and the human gut microbiome: dysbioses and accumulation of resistances." Frontiers in microbiology 6 (2016): 1543.
- Vujkovic-Cvijin, Ivan, et al. "Dysbiosis of the gut microbiota is associated with HIV disease progression and tryptophan catabolism." Science translational medicine 5.193 (2013)
- "C. difficile Infection.” Mayo Clinic, Mayo Foundation for Medical Education and Research, 18 June 2016, www.mayoclinic.org/diseases-conditions/c-difficile/symptoms-causes/dxc-20202389. Accessed 28 July 2017.
- Colledge, Helen, and Jacquelyn Cafasso. “ Pylori Infection.” Healthline, Healthline Media, 13 Oct. 2015, www.healthline.com/health/helicobacter-pylori?algo=true#causes2. Accessed 28 July 2017.
- “Microbes Help Produce Serotonin in Gut | Caltech.” The California Institute of Technology, www.caltech.edu/news/microbes-help-produce-serotonin-gut-46495. “The Human Microbiome.” Human Microbiome RSS, org/overview/about.php
- Nakatsuji,T., et al. "Antimicrobials from human skin commensal bacteria protect against Staphylococcus aureus and are deficient in atopic dermatitis." Science Translational Medicine. 9(378): eaah4680. 2017.
- Iwase, Tadayuki, et al. "Staphylococcus epidermidis Esp inhibits Staphylococcus aureus biofilm formation and nasal colonization." Nature 465.7296 (2010): 346-349.
- Holz, C., et al. "Novel bioactive from Lactobacillus brevis DSM17250 to stimulate the growth of Staphylococcus epidermidis: a pilot study." Beneficial Microbes. 8(1): 121-131. 2016 .
- Wang, Y., et al. "A Precision Microbiome Approach Using Sucrose for Selective Augmentation of Staphylococcus epidermidis Fermentation against Propionibacterium acnes. International Journal of Molecular Sciences. 17(11): 1870. 2016.
- Clavaud, C., et al. "Dandruff Is Associated with Disequilibrium in the Proportion of the Major Bacterial and Fungal Populations of Colonizing the Scalp." PLoS ONE. 8(3): e58203. 2013.
- Cision PR Newswire. https://www.prnewswire.com/news-releases/eligo-bioscience-secures-20-million-series-a-from-khosla-ventures-and-seventure-to-bring-precision-medicine-to-the-microbiome-300525473.html
- Kallyope Inc. press release (2015). https://www.kallyope.com/kallyope-inc-launches-with-44m-series-a-financing-to-harness-the-potential-of-the-gut-brain-axis/
- Xycrobe News. http://www.xycrobe.com/news.html
- Evolve Biosystems News. https://www.evolvebiosystems.com/news/
- Enterome Newsroom. http://www.enterome.fr/site/enterome-raises-e32-million-38-5-million-in-a-series-d-financing/
- Rebiotix press release. http://www.rebiotix.com/news-media/press-releases/
