How to Talk like a Medicinal Chemist in 10 minutes
I have worked in the drug discovery area for decades, and I often find that scientists, new or outside the field, to be very confused in the terminology used by medicinal chemists. Below is a quick introduction to the vocabulary used on a daily basis in drug discovery, to get you talking like a PRO in no time!
The process of development starts with Discovery. Discovery is where novel chemical compounds with biological activity are identified. This biological activity may be from the interaction with a specific enzyme or with an entire organism. These identified Hits are often found during the screening of chemical libraries, computer simulation, or screening of naturally isolated materials, such as from plants, bacteria, and fungi. A Hit Confirmation is then conducted to verify that compound is interacting with its intended biological target.
Hit to Lead, also called Lead Generation, is the process of chemically modifying the hit molecule to improve its activity towards a specific biological Target(s), while reducing undesired biological traits (such as toxicity and side-effects). Modified hits, where the chemical structure has been systematically varied, are called Analogs. Synthesizing analogs of a “Hit” is referred to as Hit Expansion. Medicinal chemists synthesize these analogs in the lab, using well-established organic chemistry techniques. To increase the synthetic throughput, chemists often focus on a specific reaction or set of reactions to assemble Building Blocks together to make a series of analogs quickly. “Building Block” is a term used to describe a compound that possesses both a reactive functional group (needed to bond with another building block) and an atom(s) that 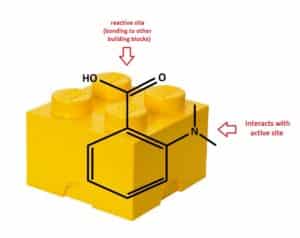
After analogs are synthesized, the biologists conduct Biological Screening of these analogs. Among other things, biochemists look at the Enzyme Activity, where one can see how well the analogs bind to the active site. Generally, the better the Binding (analog-active site attraction), the better the activity, but not always. Additionally, they may also look at the enzyme binding to undesired targets, to predict how toxic the compound may be. Biologists test in vitro (outside of a living organism) for Whole-Cell Activity, which assesses the activity of the analog on the Cellular Level. They also test on Human Cell Lines, which assesses the Toxicity of the analog.
After the screening, a meeting with medicinal chemists, computational chemists, cell biologists, microbiologists, biochemists, and intellectual property attorneys usually takes place to evaluate the next step. At which point, additional analogs are proposed, and the whole process starts again. The analogs are optimized when all parameters are met. This is when the analogs are evaluated in vivo (in a living organism), usually in mice, to test for whole animal effectiveness.
Medicinal chemists, biologists, and intellectual property attorneys work on assessing the Freedom to Operate landscape. This will tell if commercializing a product can be done without infringing on the intellectual property rights of others. If there are no patents claiming a specific idea as Intellectual Property (IP), a Utility Patent is filed to protect that idea for 20 years. This is can be a Composition of Matter Patent, where specific compound(s) is claimed. Alternatively, a Process Patent is filed, which claims a method for synthesizing specific compound(s).
The collective data will provide guidance to the upper management, and a decision for lead compound to go into the clinic or the for the whole project to be canceled (GO/NO GO Decision). If the decision is a “GO”, the “Lead Compound” is advanced into Clinical Trials, and tested In Man. The goal is to Get it Through the Clinic and get FDA Approval. At that time, it can be sold to the general population.
That was most of the terminology used in drug discovery. Now, that wasn’t so bad, was it?! Next time the topic of medicinal chemistry comes up, you’ll be prepared to show off your vocabulary!
About the author: Dr. Francavilla has a Ph.D. in chemistry, and has over 20 years of industrial experience in drug discovery, analytical development, manufacturing, and regulatory affairs. He has worked for 3 major pharmaceutical companies, and has helped get several startups to get off the ground.
