Microbiology Video: Testing Antimicrobial/Preservative Effectiveness
In this Emery Pharma microbiology video we discuss testing the effectiveness of antimicrobials and preservatives in a wide range of products.
While preservatives are commonly known for their use in foods and beverages, they’re also found in cosmetics, lotions, medications, and even eye drops. During manufacturing, these products can be exposed to microbial contamination—but contamination is more likely during actual consumer use, especially since microbes are present on our fingertips and in the air.
To address this, the United States Pharmacopeia (USP) has developed the Antimicrobial Effectiveness Test, also known as USP <51>, to evaluate how well a product prevents microbial growth over time.
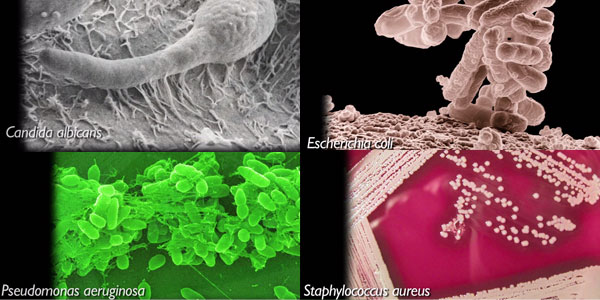
Products are challenged with a broad panel of microbes, including:
- Aspergillus brasiliensis
- Candida albicans
- Escherichia coli
- Pseudomonas aeruginosa
- Staphylococcus aureus
These microorganisms are known to cause serious, and sometimes life-threatening infections, particularly in immunocompromised individuals.
Fortunately, the European Pharmacopoeia (EP 5.1.3) and the Japanese Pharmacopoeia (JP19) have developed similar antimicrobial preservative tests, ensuring global consistency in safety standards.
To learn more, check out the blog: “Understanding Preservatives: Antimicrobial Effectiveness in Everyday Products”. You can also contact us online or call 510-899-8828 to schedule a FREE one-hour consultation.
About the Author
Originally authored by Liliana Pang. This article was reviewed and updated on June 2, 2025 by Dr. Janet Liu, current Director of Biology.
